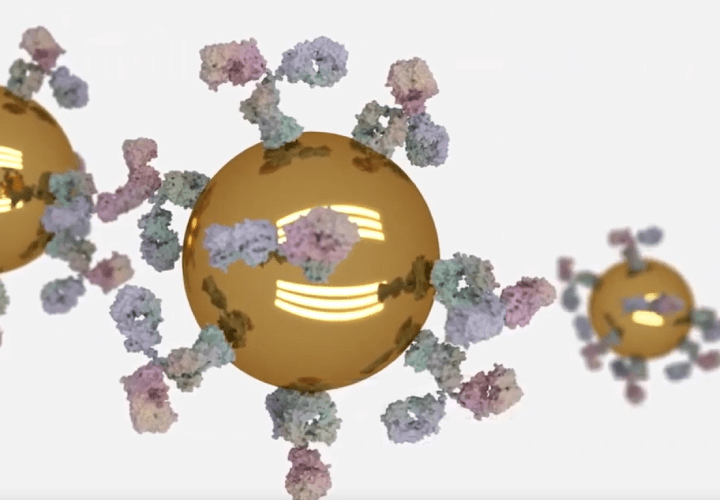
The method of conjugation is a critical element in the development and manufacture of any lateral flow assay. The potential conjugation technologies have grown over recent years as the demand for quantitative and more sensitive assays has increased. In this blog, we explore the options open to you. The first… Read More
In this article we explore the concept of integration within lateral flow product development, and what considerations are needed when planning your lateral flow project. Within lateral flow diagnostics the need to integrate development steps, ongoing processes, software systems, and supply chains are key requirements of an effective project plan… Read More

When Theresa May was UK Prime Minister, she pledged to revolutionise the health service by deploying artificial intelligence in the NHS, aiming to prevent over 20,000 cancer-related deaths each year by 2033. The Prime Minister wanted industry and charities to work with the NHS to develop algorithms that can use… Read More

Technical transfer Technical transfer is defined as the verification process by which a rapid test transfers from research and development (R&D) pilot scale production to high throughput manufacturing. At the end of the process, the product is classed as a manufactured product with all necessary validation and… Read More

In the following article, we discuss 3 important elements that ensure an assay is manufactured in a way that meets market requirements. Get the basics right Successful lateral flow manufacturing involves; conjugating the labels and antibodies effectively as well as selecting and applying the right reagents,… Read More

There are three key elements to manufacturing lateral flow assays that enable consistently accurate and reproducible results: Expertise Experience Automation Expertise ensures that an assay is optimised and scaled-up efficiently for routine manufacturing; experience means tried and tested processes and procedures are utilised, regulatory considerations are taken into account, and… Read More

Selecting reagents for a lateral flow device (LFD) is a key stage in the development of an LFD, and the selection process involves several key considerations. In this article, we highlight those key considerations. Although we mainly discuss antibodies, the following also applies to the selection… Read More
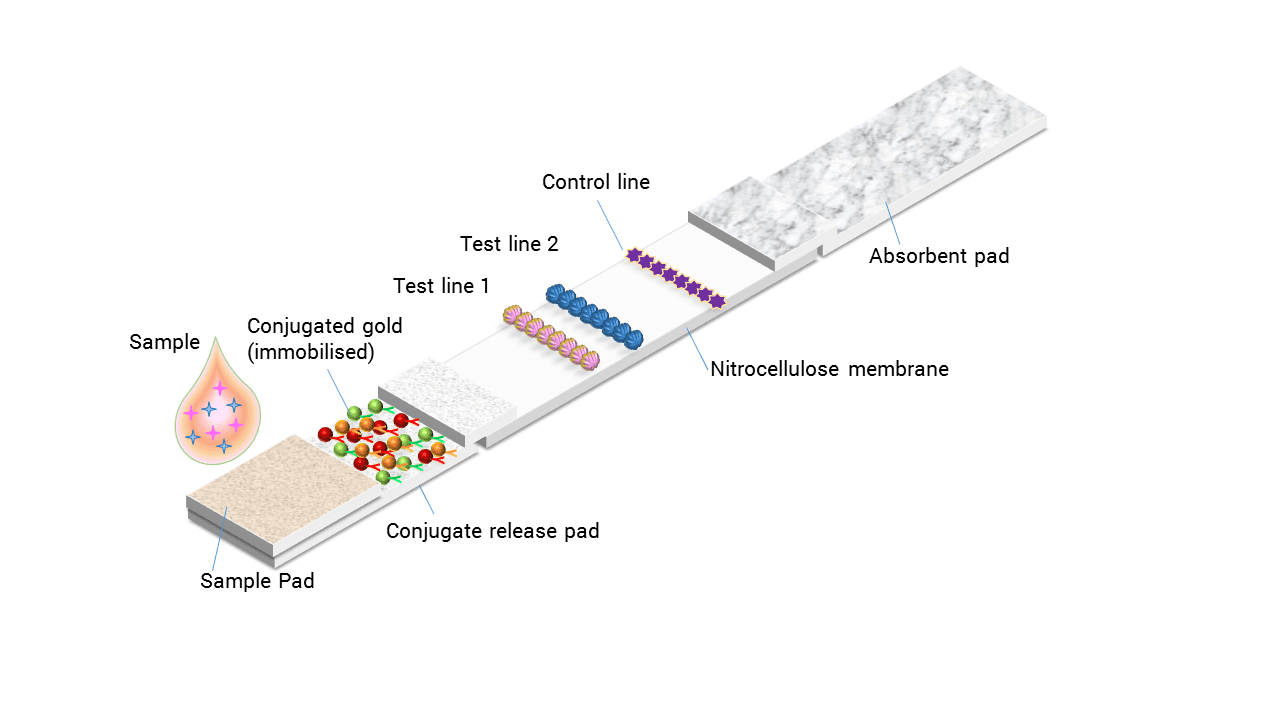
Rapid diagnostics, in essence, brings testing away from the laboratory and closer to the patient, in the case of clinical diagnostics or veterinary diagnostics, or quite literally into the field, in the case of agricultural diagnostics. One key benefit of rapid testing is faster decision making which leads to earlier… Read More
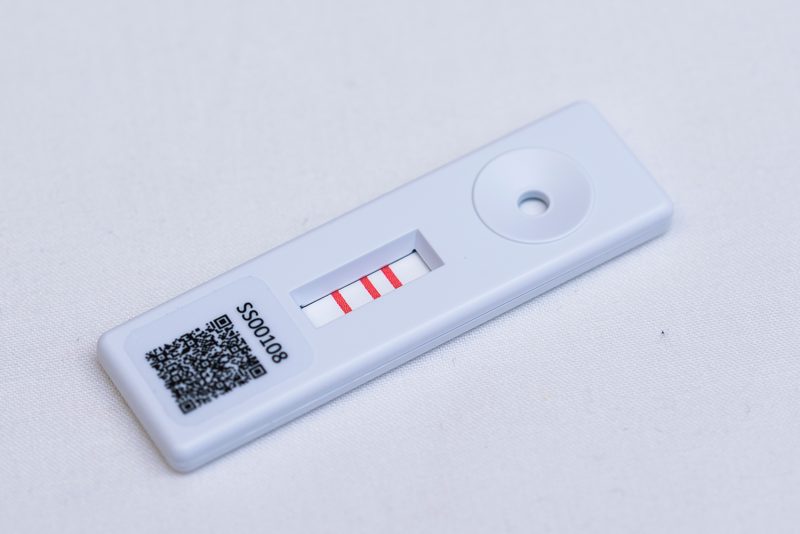
There are some common myths that lateral flow devices are inflexible and not suitable or flexible enough for an ever demanding marketplace. Therefore, we dispel some of these common myths about lateral flow devices (LFDs). Myth - LFDs can only be qualitative. In the early days,… Read More