A rapid and portable nucleic acid detection solution
Our nucleic acid lateral flow test customisation service is available to clients looking for a bespoke version of PCRD and PCRD Flex. Alternatively, clients with existing nucleic acid tests can work our high-volume manufacturing team to meet spikes in demand or accelerate growth plans.
In-house regulatory support and many years of lateral flow experience alongside one of Europe’s largest rapid test manufacturing capacities means we can meet all market and volume needs.
Our process
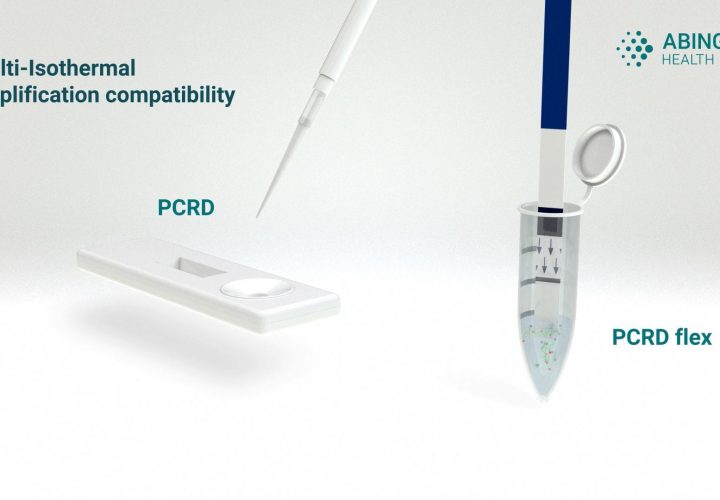
Bulk purchase of our standard PCRD and PCRD Flex is available. Alternatively, these nucleic acid lateral flow immunoassays (NALFIA) can be configured to meet specific specification requirements.
Flexible nucleic acid test development and manufacture for all specification and volume needs.
- Development and manufacture tests for both single and multiple analytes
- Flexible and scalable solutions in line with growth plans
- Adaptability in response to spikes in demand
- Cassette or dipstick versions and multiple kitting options
- Stable pricing for budget planning
- International shipping and logistics
- In-house regulatory and commercialisation support

How To Use
PCRD and PCRD FLEX are easy to use and complement most isothermal amplification techniques.
Click arrows on both sides to scroll
Related Services
Explore our full suite of flexible, industry leading lateral flow rapid test services
Knowledge Centre
Learn more about lateral flow technology and Abingdon Health’s development and manufacturing expertise.






